Organizations using both a RIM Vault and a PromoMats Vault can utilize the Spark messaging framework to create a standard Vault to Vault connection.
The RIM-PromoMats Connection’s AdPromo Submission Management feature enables a seamless flow from compliance package generation in PromoMats to Content Plan creation in RIM Submissions, and, if licensed, to the FDA through the gateway via RIM Submissions Publishing, reducing the time required to submit the compliance package to the FDA. This connection transfers Application, Submission, and Compliance Package information across Vaults and automates the creation and updating of CrossLink documents.
Note: The RIM PromoMats Connection’s AdPromo feature is only available for organizations with both a RIM Submissions Vault and PromoMats Vault.
The RIM-PromoMats Connection’s Product Data feature allows the transfer of Product, Brand, and Registration information needed within Regulatory and Commercial business teams.
Vault RIM is the source of truth for most product data needed within Commercial Vaults. Product Family, Product (Product Form), Product Variant, Packaging, Therapeutic Indication information is transferred from RIM Vaults to PromoMats.
Brand records are created and managed in PromoMats. Brand (Trade Name) information is transferred from PromoMats Vaults to the Regulatory Text object within RIM Vaults, using the type of Drug Trade Name or Device Trade Name.
For organizations with Registrations, RIM also provides the source of truth for registrations data needed within Commercial Vaults. Registration, Registered Regulatory Text (Registered Brand), Registered Package Product & Registered Indication information is transferred from RIM Registrations to PromoMats.
Note: The RIM-PromoMats Connection’s Product Data feature is only available for organizations with both any RIM Submissions Vault and a PromoMats Vault, although the Registrations related data is only available to organizations with Registrations.
About Record Creation & Update
Vault creates new records in their lifecycles’ starting states. When creating a record through the connection, Vault also populates the Link (link__sys) field on the target record with the source record’s Global ID (global_id__sys). These fields let Vault know which records to update in the target Vault when data is updated in the source Vault.
When users delete documents or object records in one Vault, Vault does not delete the associated documents or object records in the connected Vault.
About the RIM-PromoMats AdPromo Submission Management Feature
When your RIM Submissions and PromoMats Vaults are first connected and the AdPromo Submission Management feature is configured, Vault creates any existing RIM US Application in PromoMats that has a Last Modified Date that is after the Last Successful Run date of the integration. Any Applicant records linked to these Applications are also created. To prevent duplicate records, Vault does not create records if the Link fields match. Vault also does not create any new Applicant or Application records from RIM with the same Name field value but different Global IDs as existing records in PromoMats. To ensure that Vault recognizes that these records match, you’ll need to copy the Global ID for the record in RIM and add it to the Link field in PromoMats.
After connecting your Vaults, you must move at least one compliance package to the Submission Ready state. This triggers RIM to pull all Submission Ready eCTD binders from PromoMats to RIM. This eCTD binder must use an Application and Applicant record that is linked to an Application and Applicant record in RIM.
When an eCTD Compliance Package moved to the Submission Ready state, this triggers the connection to:
- Create a Submission record in your RIM Vault, appending the PromoMats Package record’s Global ID to the record’s Name to ensure records are unique, for example “Compliance Package Name - 123_101”, where the Compliance Package’s Global ID is
123_101. - Copy the Submission record in PromoMats and add the record to the eCTD binder and corresponding documents
- Create a Content Plan in RIM, if configured.
- Vault sets the Published Output Location for all matched documents in the submission content plan to maintain the relative path links constructed in PromoMats. The Published Output Location the connection sets may differ from what is configured in the content plan template.
- Create CrossLinks in RIM to the documents in PromoMats.
- Existing CrossLinks in RIM do not reflect metadata updates to documents within the PromoMats eCTD compliance package. This behavior is to preserve any values used for submission content plan activation and document automatching.
- Activate continuous publishing
- When publishing source documents or renditions, Vault references the Source for Published Document field. Vault sets the Source for Published Document field to Source for Content Plan Items that have an XML Leaf Attribute Type of Form Type to prevent the flattening of fillable forms when a rendition is used instead of the source document. See Working with Submissions Publishing for more details.
About Document Creation & Integrated CrossLinks
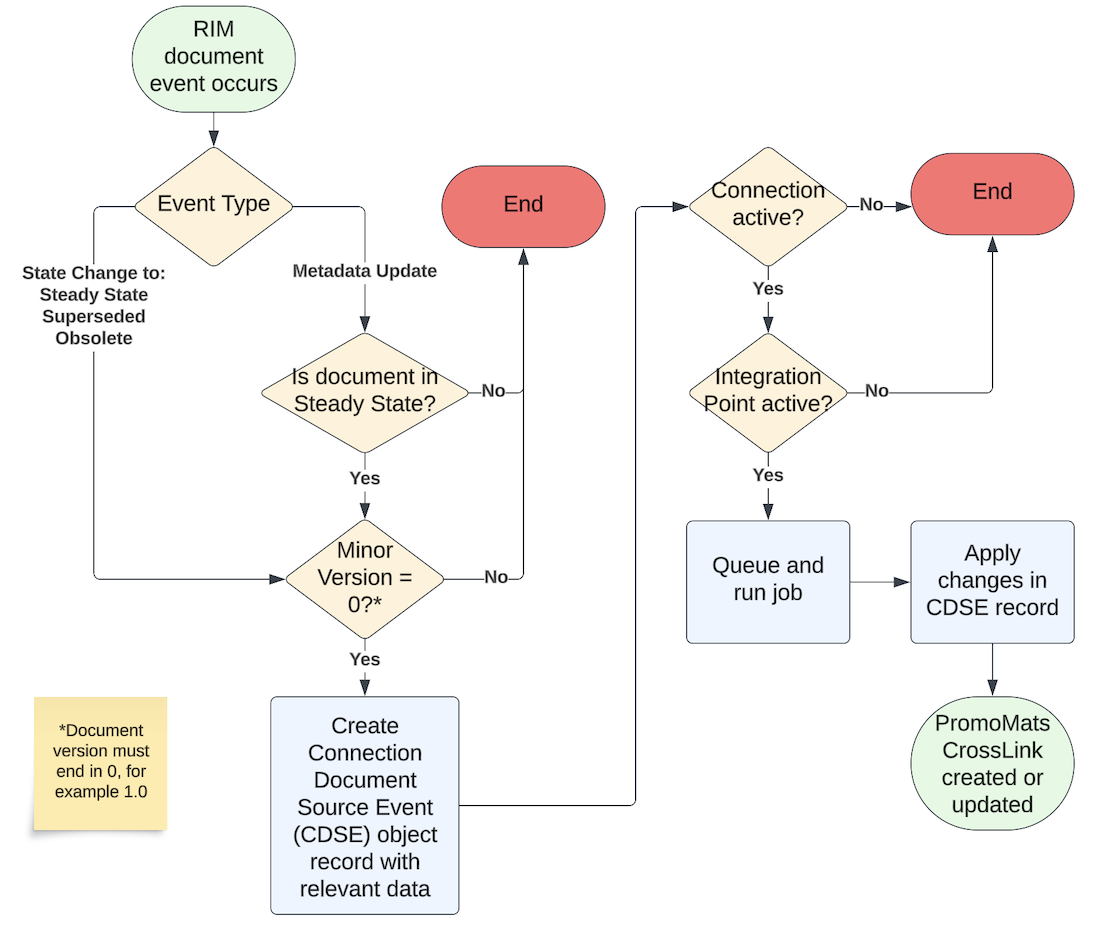
The continual availability of RIM documents in PromoMats can be automated using Admin-configured Integrated CrossLinks. With this capability enabled, Vault creates new CrossLink documents in the steady state defined in the target Vault’s document lifecycle of a given document type.
Integrated CrossLinks create and maintain PromoMats CrossLink documents when a RIM document:
- Enters the Steady state for the first time. This is the default connection behavior which creates the PromoMats CrossLink.
- Undergoes a metadata update while in the Steady state.
- Re-enters the Steady state after revision.
- Enters the Superseded or Obsolete state.
This feature automatically uses the Specific Document Version binding rule only, ensuring users can add link annotations and anchors in PromoMats to documents originally authored in RIM.
The diagram below provides a basic overview of this process, as well as the various criteria which must be met in order for a RIM document to be CrossLinked and maintained in PromoMats. See also Integrated CrossLink Limitations for more information.
Integrated CrossLink Examples
VeePharm classifies master prescribing information in RIM under the Labeling > Product Information > Prescribing Information document classification, and in PromoMats under the Reference > FPI - Full Prescribing Information document subtype.
These documents’ respective lifecycles map state types to states as follows:
| State Type | RIM State | PromoMats State |
|---|---|---|
| Steady State | Current In-Use | Approved for Use |
| Superseded State | Superseded | Superseded |
| Obsolete State | Historical | Expired |
The table below illustrates Vault actions and outcomes for a given prescribing information document as it undergoes each document event.
| RIM document event for Prescribing Information document | PromoMats result for FPI - Full Prescribing Information CrossLink |
|---|---|
| RIM draft document v0.1 routed for approval, entering the Approved for Use state v1.0. | None. RIM’s Approved for Use state is not the Steady state, therefore a CrossLink is not created. |
| RIM document v1.0 moved to the Current In-Use (Steady) state. | PromoMats CrossLink v1.0 created in the Approved for Use (Steady) state. |
| RIM document v1.1 created and routed for approval. It enters the Approved for Use state (v2.0), then the Current In-Use (Steady) state. Document v1.0 set to the Superseded state. | PromoMats CrossLink updated to v2.0, remaining in the Approved for Use (Steady) state. CrossLink v1.0 set to the Superseded state. |
| RIM document v2.0 Country document field updated from “United States” to “Puerto Rico”. | PromoMats CrossLink v2.0 Country document field updated from “United States” to “Puerto Rico”. |
| RIM document v2.0 moved to the Historical (Obsolete) state. | PromoMats CrossLink v2.0 moved to the Expired (Obsolete) state. |
These examples mostly follow a positive flow, in that document events are encountered and processed as expected. Below are some possible scenarios in which a new or updated RIM document produces different results in PromoMats.
| RIM document event for Prescribing Information document | Scenario | PromoMats result for FPI - Full Prescribing Information CrossLink |
|---|---|---|
| RIM document v1.0 up-versioned in quick succession to Current In-Use (Steady) state v2.0, then v3.0 (Steady state). | Rapid transition to v3.0 occurs before the connection job processes the transition to v2.0. | PromoMats CrossLink v1.0 up-versioned to Approved for Use (Steady) state v3.0. Version history reflects versions 1.0, 2.0, and 3.0. |
| RIM document v3.1 Country document field updated from “United States” to “Puerto Rico”. | RIM document v3.1 is in the draft state. | PromoMats CrossLink v3.0 Country field remains as “United States” until the RIM document enters the Steady state (v4.0) and Vault updates the CrossLink document and metadata. |
| RIM document v3.0 up-versioned to Current In-Use (Steady) state v4.0. | PromoMats CrossLink v3.0 exists, however the PromoMats Superseded state type does not have a mapped state. | PromoMats CrossLink v3.0 remains in its current Approved for Use (Steady) state. User Exception Message generated. |
| RIM document v3.0 moved to the Historical (Obsolete) state. | PromoMats CrossLink v3.0 exists, however the PromoMats Obsolete state type does not have a mapped state. | PromoMats CrossLink v3.0 remains in its current Approved for Use (Steady) state. User Exception Message generated. |
Transferred Data & Documents
| Object or Documents | Source Vault | Target Vault |
|---|---|---|
| Applicant | RIM (Vault Submissions) | PromoMats |
| Application | RIM (Vault Submissions) | PromoMats |
| Brand/Regulatory Text | PromoMats | RIM (all applications) |
| Documents | RIM (Vault Submissions) | PromoMats |
| Product Hierarchy | RIM (all applications) | PromoMats |
| Registration Hierarchy | RIM (Vault Registrations) | PromoMats |
| Therapeutic Indication | RIM (all applications) | PromoMats |
Label Management
When the Connection creates the Submission record and Content Plan for AdPromo submissions in RIM, it also determines if the label in the Compliance Package is a document that originated in RIM, and if that label has been previously submitted to the Application.
The label’s origin determines the system-managed steps as follows:
| Scenario | Connection Automated Steps |
|---|---|
| 1. Label in the Compliance Package is not a CrossLink that originated in RIM | Create a CrossLink document in RIM, match CrossLink to Content Plan |
| 2. Label in the Compliance Package is a CrossLink that originated in RIM, AND the label was previously submitted via Veeva RIM Publishing | Identify source label in RIM, AND Create a Reference Leaf in Content Plan |
| 3. Label in the Compliance Package is a CrossLink that originated in RIM, AND the label was not previously submitted via Veeva RIM Publishing | Identify source label in RIM, AND match source label to Content Plan |
| 4. Label in the Compliance Package is a CrossLink that originated in RIM, BUT the source was deleted, or is an Obsolete/Superseded state | Document is not matched to Content Plan, AND User Exception Message is generated |
When Veeva RIM Publishing is enabled, Vault searches Submission metadata to determine where the label was used. The search priority order for previously submitted documents is:
- Submissions within the same Primary Application.
- Submissions with the same
lead_market__v(_Country_),health_authority_division__v, and only where the Allow Cross Application Linking field = TRUE for thelead_market__v. - Last Modified Date
Matching the source document prevents the connection from creating a circular CrossLink to a CrossLink for labels that originated in RIM and were returned to RIM via the Compliance Package transfer. Cross-application Reference Leafs are also supported for applications where the Allow Cross Application Linking option is enabled.
Note: In order for the Reference Leaf to be created in the Content Plan, the Content Plan Item’s Name field cannot be required as unique, or a User Exception Message will occur. Admins should check this configuration before utilizing this feature.
About the RIM-PromoMats Product Data feature
The RIM-PromoMats Connection’s Product Data feature allows the transfer of Product, Brand and Registration information between RIM and PromoMats to facilitate common terminology across Vaults. When the feature is configured in both Vaults, the following data points are transferred.
| When the following record is created in RIM | A corresponding record is automatically created in PromoMats |
|---|---|
Product Family (product__v) |
Product Family (product_family__v) |
Product (drug_product__v) |
Product Form (product_form__v) |
Product Family Product (product_pharmaceutical_product__rim) |
Product Family Product Form (product_family_product_form__v) |
Product Variant (product_detail__v) |
Product Variant (product_variant__v) |
Product Component (product_component__rim) |
Product Component (product_component__v) |
Packaging (packaging__rim) |
Packaging (packaging__v) |
Therapeutic Indication (therapeutic_indication__v) |
Indication (indication__v) |
| When the following record is created in PromoMats | A corresponding record is automatically created in RIM |
|---|---|
Brand (product__v) |
Regulatory Text (regulatory_text__v) |
If Registrations is licensed in your RIM Vault, the following data points can be transferred from RIM to PromoMats
| When the following record is created in RIM | A corresponding record is automatically created in PromoMats |
|---|---|
Registration (registration__rim) |
Registrations (registration__v) |
Registered Regulatory Text (registered_regulatory_text__v) |
Registered Brand (registered_brand__v) |
Registered Packaged Medicinal Product (registered_packaged_medicincal_product__v) |
Registered Packaged Product (registered_packaged_product__v) |
Registered Indication (registered_indication__rim) |
Registered Indication (registered_indication__v) |
Managing User Exception Messages
You may see a User Exception Message when using this feature. Navigate to Admin > Configuration > Objects > User Exception Message Items. Go to Layouts, and Insert a Section for a Related Object. Select User Exception Item as the object. Click Edit and set the Display values in Business Admin menu checkbox, then click Save. Then, navigate to Business Admin > Objects > User Exception Messages to view the message details. To view specific records or documents that have encountered an error, navigate to Admin > Connections > User Exception Messages, click on thr Exception, and view specific items in the User Exception Items section. After the issues are resolved, restart rerun the connection by going to Admin > Connections > User Exception Message and clicking Rerun Integration on the message record.